Research
Major Research Experiences
1. Structural Diversity of Mammalian Motile Cilia
- Developed a novel protocol for isolating swine ependymal and bovine oviduct cilia.
- Used single-particle cryo-EM to resolve high-resolution structures of mammalian motile cilia doublet microtubules.
- Uncovered structural diversity across mammalian axonemes.
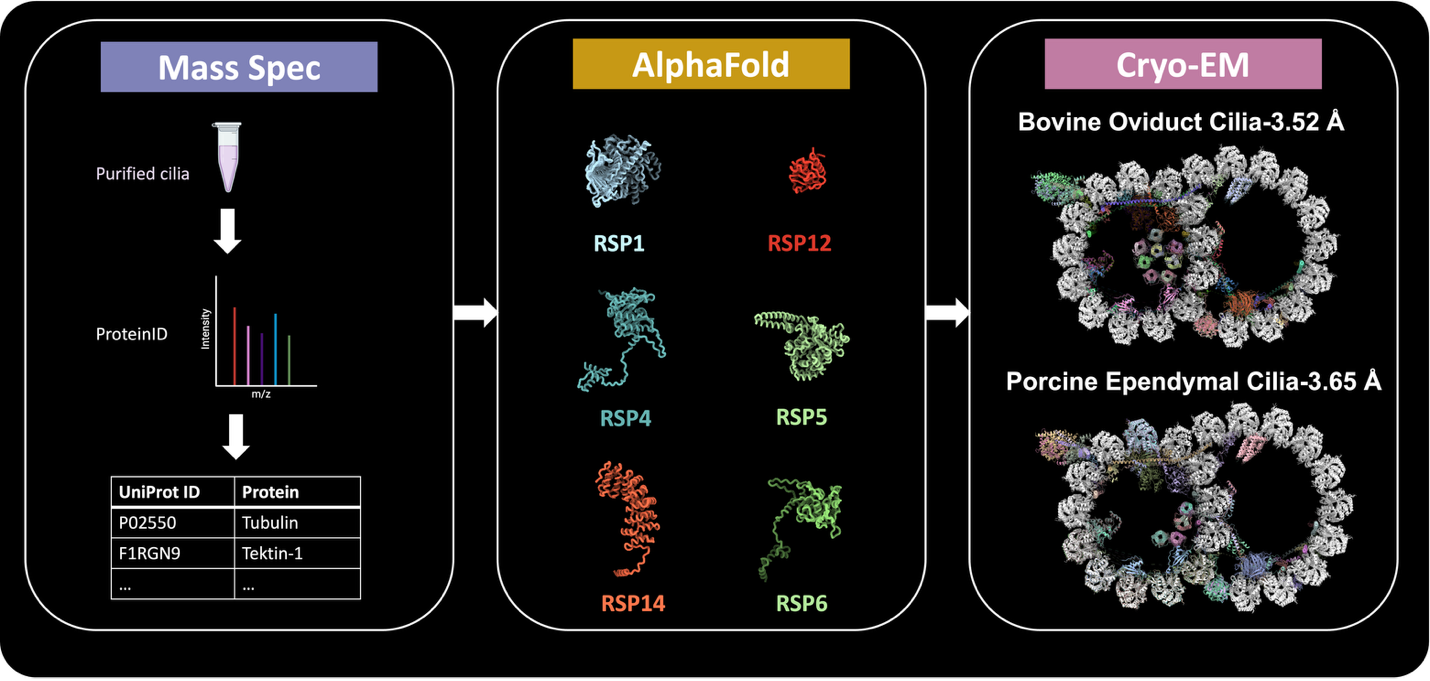
Figure 1. AI-assisted protein identification workflow.
2. Human endogenous retrovirus K (HERV-K) envelope structures in pre- and post-fusion
First Structural Characterization
This study reports the first high-resolution cryo-EM structures of the HERV-K envelope (Env) glycoprotein in both pre-fusion (2.2 Å) and post-fusion (2.8 Å) conformations.Novel Monoclonal Antibodies
A panel of ten novel monoclonal antibodies was developed and characterized to recognize distinct subunits and conformational states of HERV-K Env. Two antibodies, Kenv-6 and Kenv-4, facilitated structure determination.Pre-fusion Env Architecture
The pre-fusion Env is an elongated trimer with SU subunits forming an inverted tripod and TM subunits forming a clasp underneath. This structure is distinct from all known retroviral Env structures.Engineering a Stable Pre-fusion Trimer
Successful stabilization of the metastable pre-fusion Env trimer was achieved using structure-guided mutations: a disulfide bond, furin site modification, and trimerization domain fusion.Unique TM Features in Post-fusion State
The post-fusion TM forms a typical six-helix bundle but includes a unique “tether helix” not observed in other retroviral Envs, indicating structural divergence.SU Fold is Unique Among Retroviruses
The SU subunit adopts a novel fold with a β-sheet-rich architecture, showing no structural homology to HIV-1, SIV, or Syncytin-2 SU, except for a conserved β-sheet in the base domain.Antibody Epitope Mapping
Kenv-6 binds a conserved, conformational epitope at the SU apex, while Kenv-4 recognizes a post-fusion TM epitope spanning two protomers. These mAbs are valuable tools for detecting native Env.Biological and Therapeutic Relevance
HERV-K Env is aberrantly expressed in various cancers and autoimmune diseases. The described structures and antibodies provide a foundation for developing diagnostics and immunotherapies.Glycan Shield Analysis
Env carries 10 N-glycans per monomer, providing moderate shielding (31% on SU, 57% on TM). It is less shielded than HIV-1 Env, potentially exposing vulnerable epitopes for immune targeting.Implications for Disease and Therapy
Findings suggest the structural basis for HERV-K Env’s role in disease and highlight its potential as a therapeutic target for cancer and autoimmune conditions.
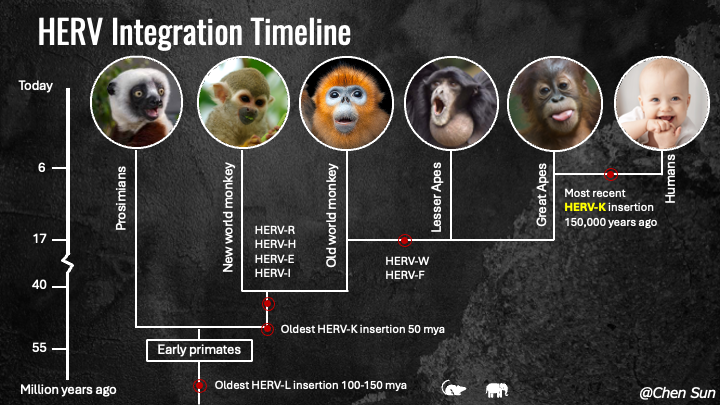
Figure 2. HERV integration timeline.
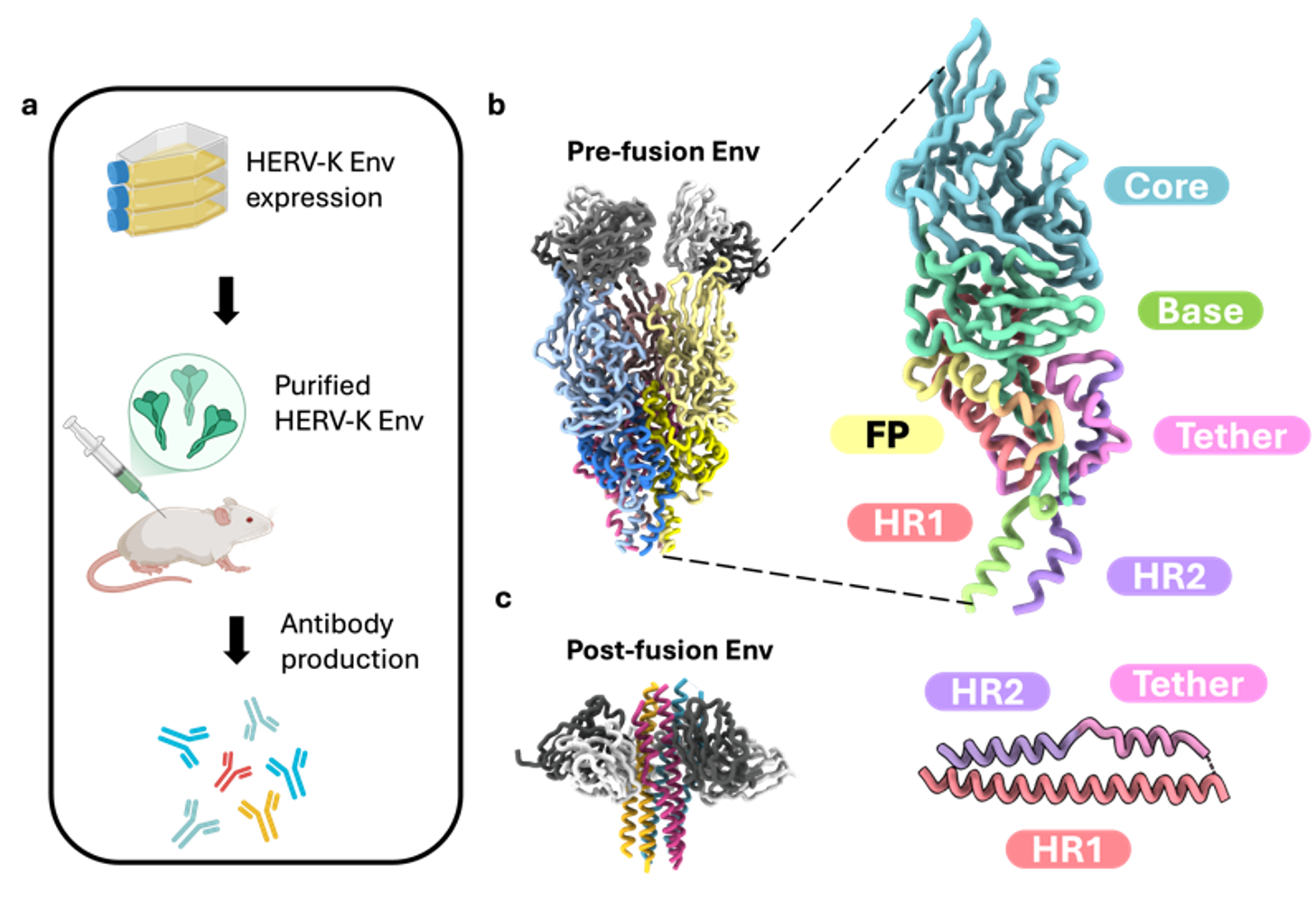
Figure 3. Antibody assisted structure determination of HERV-K env in pre- and post- fusion conformations.
3. 3D Molecular Architecture of CAR T Cell Immune Synapses Revealed by Cryo-ET
- Investigated immune synapse formation between CAR T cells and cancer cells using cryo-electron tomography.
- Revealed key structural features enhancing our understanding of CAR T cell-mediated cytotoxicity.
4. Tulane Virus Structure and Receptor Switch
- Studied a novel Tulane virus variant with abolished receptor binding due to minor capsid protein mutations.
- Developed an innovative virus purification method, significantly enhancing yield and purity.
- Resolved the structure of the variant at 2.6 Å, revealing the structural basis for the receptor switch.
5. Method Development in Single-Particle Cryo-EM
- Developed a web app for real-space helical indexing, enabling easy and reliable determination of helical parameters. Check out HI3D.
- Addressed air-water interface challenges by using encapsulin-based encaging.
- Optimized data collection strategies for the Volta phase plate to improve efficiency.
